Medical Accelerators
Lioness MedTech promote and service leading MedTech within radiotherapy
Lioness Medtech promote and service leading medical technologies to diagnose and treat disease with radiation. Only the very best technologies.
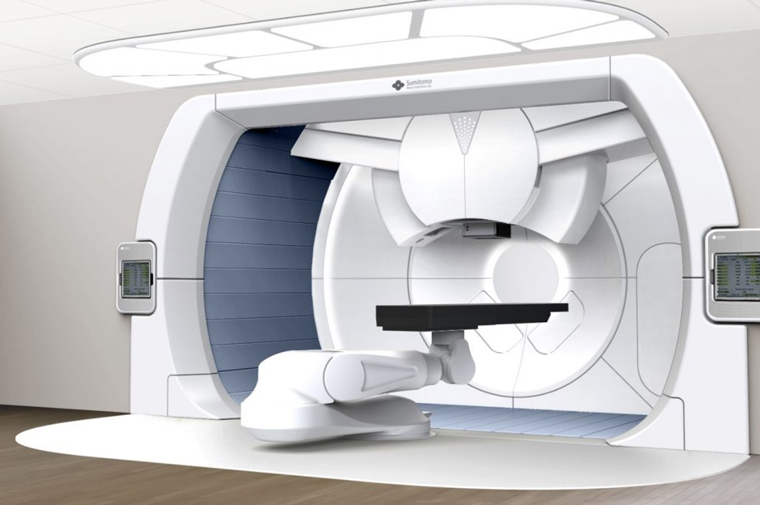
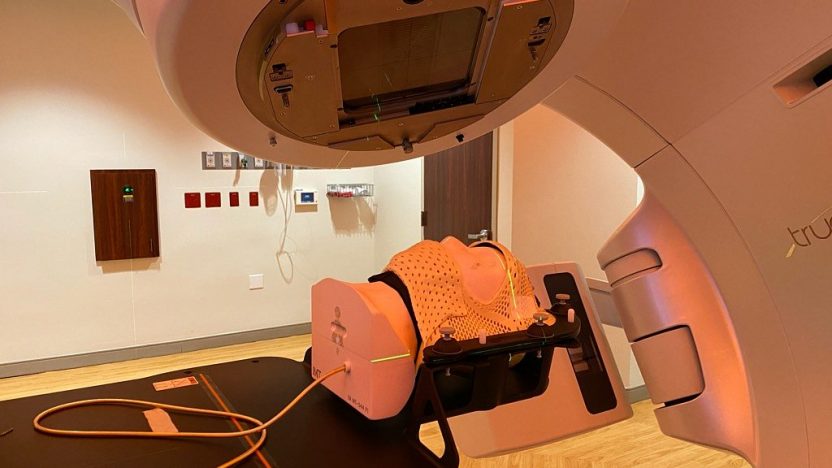
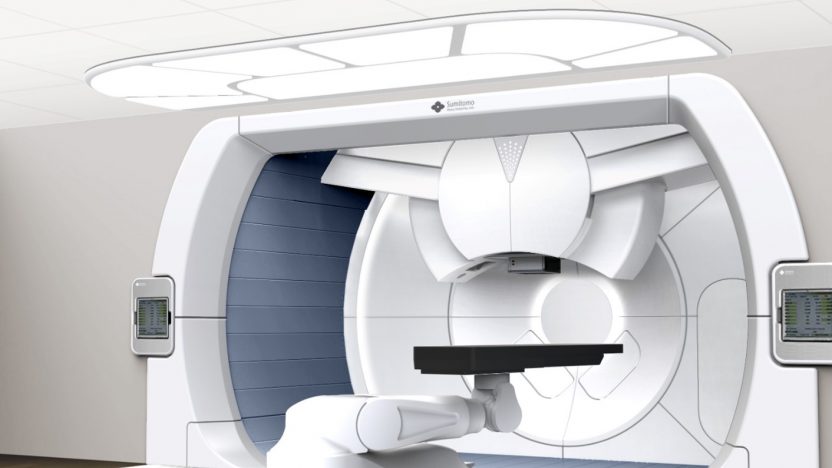
Proton Therapy, PET Cyclotrons, BNCT and light ions particle therapy: Lioness MedTech represent Sumitomo Heavy Industries Ltd. as European agents
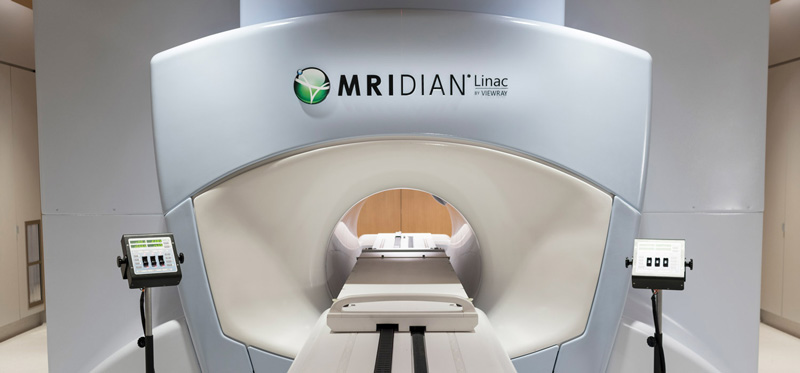
MRI-Linac: Lioness MedTech are the Nordic distributors for the MRIdian Linac from ViewRay
Stereotactic Radiosurgery of the brain (SRS): Lioness MedTech distribute ZAP-X in the Nordics
QA phantoms and software to track QC: Lioness MedTech distribute IMT (IMTQA) in the Nordics
Patient positioning and bolus: Lioness MedTech distribute AnatGe and MoldCare in the Nordics
Dreamclinic Survey
Please do our Dreamclinic Survey with 9 short questions about your future dreamclinic.